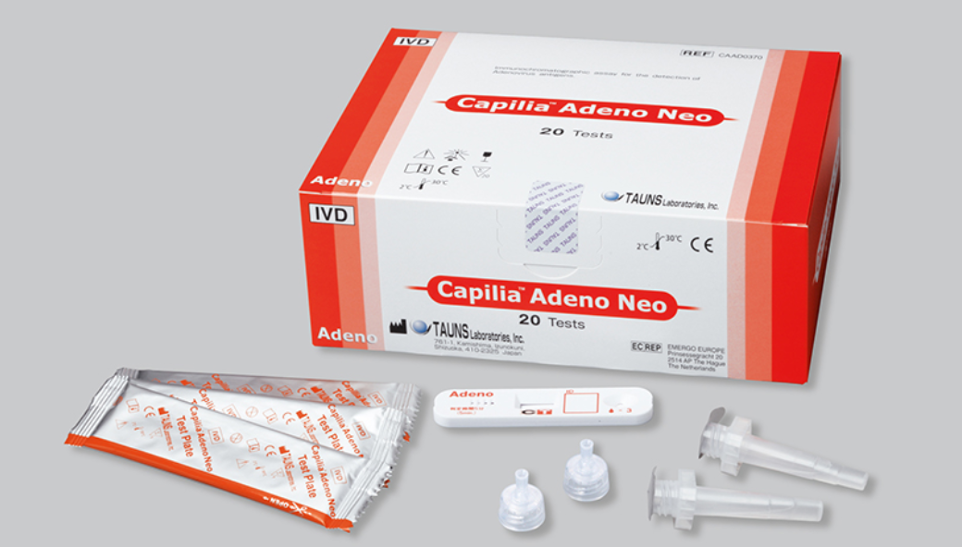
Capilia™ Adeno Neo
Capilia™ Adeno Neo is an in vitro diagnostic rapid test kit designed for the qualitative detection of adenovirus antigens in nasopharyngeal aspirate (NPA) or nasopharyngeal swabs (NPS) samples. It offers a convenient and reliable method for diagnosing adenovirus infections, which are a common cause of respiratory illnesses, particularly in children.
Capilia™ Adeno Neo: A Rapid and Accurate Adenovirus Antigen Detection Solution
Product Overview
Capilia™ Adeno Neo is a revolutionary in vitro diagnostic rapid test kit designed for the qualitative detection of Adenovirus antigens in nasopharyngeal aspirate (NPA) or nasopharyngeal swabs (NPS) samples. This cutting-edge tool empowers healthcare professionals to make timely and informed treatment decisions, effectively combating Adenovirus infections and safeguarding patient well-being.
Key Features
- Rapid and Accurate Detection: Capilia™ Adeno Neo delivers results within a remarkable 3-5 minutes, enabling prompt identification of Adenovirus infections.
- Enhanced Sensitivity and Specificity: Utilizing advanced platinum-gold colloid nanotechnology, the test exhibits exceptional sensitivity and specificity, ensuring confidence in test outcomes.
- User-Friendly Design: The test procedure is remarkably simple and straightforward, requiring minimal training and time to perform.
- Clear and Unambiguous Results: The presence or absence of Adenovirus antigens is clearly indicated by distinct black lines, eliminating ambiguity in interpretation.
- Versatile Applicability: Capilia™ Adeno Neo can be used to analyze both NPA and NPS samples, catering to a wider range of clinical settings.
Benefits
- Improved Patient Care: Timely diagnosis of Adenovirus infections facilitates appropriate treatment and management, leading to better patient outcomes.
- Effective Infection Control: Early identification of Adenovirus cases enables effective infection control measures to prevent the spread of the virus.
- Optimized Healthcare Resource Utilization: Prompt diagnosis and treatment can minimize the severity of Adenovirus infections, potentially reducing hospitalizations and healthcare costs.
Applications
- Healthcare Settings: Clinicians in hospitals, clinics, and urgent care centers can utilize the rapid Adenovirus antigen test to make informed treatment decisions and implement infection control strategies.
- Home Testing: Healthcare providers may recommend home-based Adenovirus testing for high-risk individuals, such as infants and young children, to facilitate early diagnosis and intervention.
- Surveillance and Research: Public health authorities and researchers can employ the rapid Adenovirus antigen test for surveillance purposes to monitor Adenovirus activity and gather valuable data for epidemiological studies.
Conclusion
Capilia™ Adeno Neo stands as a testament to the power of innovative technology in healthcare. Its rapid, accurate, and user-friendly design empowers healthcare professionals to combat Adenovirus infections effectively, safeguarding patient well-being and optimizing healthcare resource utilization.
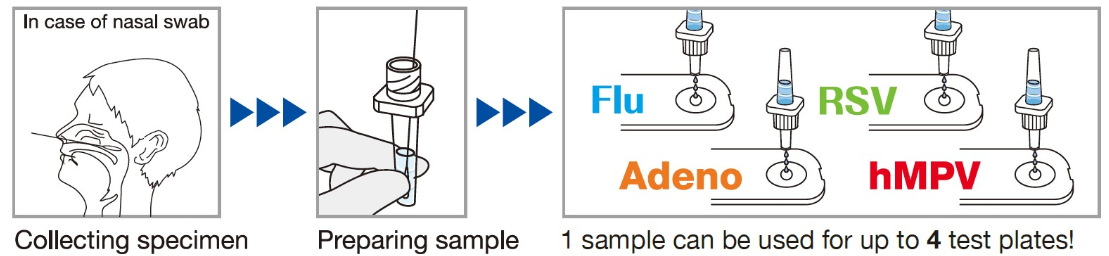
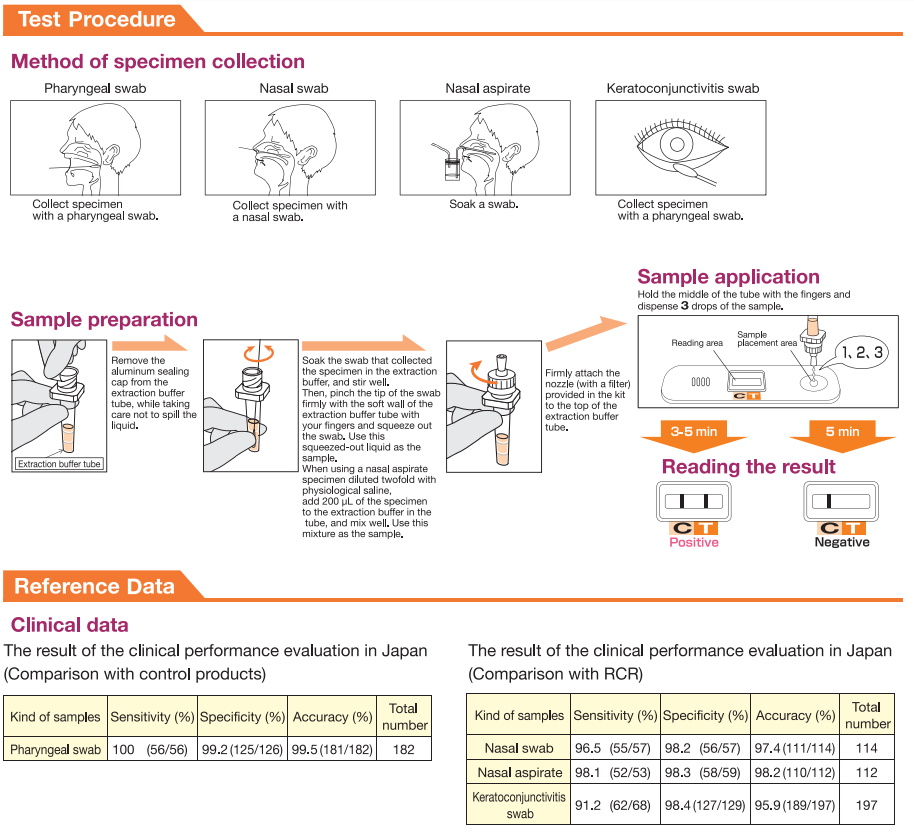
SPECIMEN COLLECTION AND PREPARATION
1) Sampling of pharyngeal swab
Firmly insert a pharyngeal swab into the pharynx
through the oral cavity, and collect the mucosal
epithelium by swabbing the posterior wall of the
pharynx and the palatine tonsil several times,
centering around the rubefacient portion. Avoid
touching saliva. If the specimen is mixed with
saliva, the test result lines may become fainter on
the test plate.
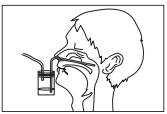
3) Sampling of nasal aspirate
Firmly insert one tube of the suction trap into
the suction pump, and the other tube into a nasal
cavity through an external nostril. Collect the nasal
discharge aspirate in the suction trap by operating
the suction pump. Soak a swab in the nasal aspirate
collected by the trap, and let the swab absorb the
nasal aspirate well. When nasal aspirate is taken
using a micropipette or other instruments, dilute
the nasal aspirate twofold with physiological saline
and sample 200
μL of this dilution.
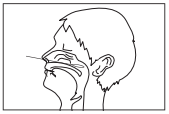
2) Sampling of nasal swab
Firmly insert a nasal swab into the nasal cavity and
collect mucosal epithelium by swabbing the nasal
turbinate several times.
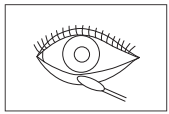
4) Sampling of keratoconjunctivitis swab
Use a pharyngeal swab, and collect the cuticle by
scratching hard the keratoconjunctivitis several
times. If necessary, use a surface anesthetic and
scratch the area of inflammation as strongly as you
can.
Adenovirus Rapid Test Kit
Intended Use
This product is a rapid immunochromatographic assay for the qualitative detection of adenovirus antigen in nasopharyngeal swabs. It is intended for in vitro diagnostic use only.
Clinical Significance
Adenoviruses are a common cause of respiratory infections, including the common cold. They can also cause conjunctivitis (pink eye) and gastroenteritis (diarrhea). Adenoviruses are most common in children under the age of 5 and in people with weakened immune systems.
Test Principle
This test utilizes a monoclonal antibody to capture adenovirus antigen present in the specimen. The antigen-antibody complex then binds to a second antibody labeled with colloidal gold particles. If adenovirus antigen is present in the specimen, a visible band will appear at the "T" line. A control band will always appear at the "C" line to indicate proper test performance.
Kit Components
- Test cassettes (individually sealed in foil pouches)
- Desiccant sachets
- Sample extraction tubes
- Sample extraction buffer
- Nozzles with filters
- Sterile swabs
- Instructions for Use
Storage and Stability
Store the kit at 2-8°C. Do not freeze. The test cassettes are stable for 24 months when stored as directed.
Specimen Collection
- Collect a nasopharyngeal swab using a sterile swab.
- Insert the swab into the patient's nostril and gently rotate it against the back wall of the nasopharynx.
- Remove the swab and place it into the sample extraction tube.
Test Procedure
- Open the foil pouch and remove the test cassette.
- Add 4 drops of sample extraction buffer to the sample extraction tube.
- Squeeze the sample extraction tube to release the sample into the buffer.
- Remove the nozzle with filter from the sample extraction tube and place it on the sample port of the test cassette.
- Add 3 drops of the sample-buffer mixture to the sample port.
- Start the timer.
- Read the results after 15 minutes.
Interpretation of Results
- Positive: A visible band appears at both the "T" and "C" lines.
- Negative: Only a visible band appears at the "C" line.
- Invalid: No visible band appears at the "C" line. Repeat the test.
Limitations
- This test is for qualitative detection only and does not provide quantitative results.
- A negative result does not rule out the possibility of adenovirus infection.
- The test may give false positive results due to cross-reactivity with other viruses.
Precautions
- Use the test kit immediately after opening the packaging.
- Wear gloves and eye protection when handling specimens.
- Dispose of used test kits and specimens properly.
Why Choose GENTAUR as Your Distributor
GENTAUR is a leading global distributor of innovative diagnostic solutions, including Capilia™ Adeno Neo. With a proven track record of excellence and a commitment to customer satisfaction, GENTAUR is the ideal partner for healthcare providers seeking the best possible solutions for Adenovirus detection.
Here are some of the reasons why GENTAUR is the best distributor for Capilia™ Adeno Neo:
- GENTAUR's extensive experience and expertise in the diagnostic industry
- GENTAUR's commitment to providing high-quality products and services
- GENTAUR's strong network of healthcare providers
- GENTAUR's dedication to customer satisfaction
When you choose GENTAUR as your distributor for Capilia™ Adeno Neo, you can be confident that you are getting the best possible product and service. GENTAUR is committed to helping you make a positive impact on patient care.
Conclusion
Capilia™ Adeno Neo stands as a testament to the power of innovative technology in healthcare. Its rapid, accurate, and user-friendly design empowers healthcare professionals to combat Adenovirus infections effectively, safeguarding patient well-being and optimizing healthcare resource utilization. With GENTAUR as your distributor, you can be confident that you are getting the best possible support for this groundbreaking product.